
One of the most frustrating problems in IVF today is that of poor ovarian response. This is also known as poor ovarian function, poor ovarian reserve, occult ovarian failure, or the oopause.
It’s well known that pregnancy rates in IVF are directly dependent upon the quality and number of embryos transferred, and the more the eggs a woman grows, the better the embryos we can select from. This is why women with a good ovarian response have much higher pregnancy rates than women with a poor ovarian response.
Usually, ovarian functions goes hand in hand with age, and as a women becomes older, her ovarian response starts declining. Every girl is born with a finite number of eggs, and their number progressively declines with age. A measure of the remaining number of eggs in the ovary is called the "ovarian reserve"; and as the woman ages, her ovarian reserve gets depleted. The infertility specialist is really not interested in the woman's calendar (or chronological age), but rather her biological age - or how many eggs are left in her ovaries.
Various tests have been described to measure ovarian reserve. The commonest test measures the level of FSH ( follicle stimulating hormone) in the blood - the basal ( day 3) FSH level. A high level suggests poor ovarian reserve; and a very high level ( more than 20 mIU/ml, though this varies from lab to lab ) is diagnostic of ovarian failure. A test that can provide earlier evidence of declining ovarian function is the clomiphene citrate challenge test ( CCCT).
This is similar to a " stress test " of the ovary; and involves measuring a basal Day 3 FSH level; and a Day 10 FSH level, after administering 100 mg of clomiphene citrate from Day 5 to Day 9. If the sum of the FSH levels is more than 25, then this suggests poor ovarian function, and predicts that the woman is likely to have a poor ovarian response ( she will most probably grow few eggs, of poor quality) when superovulated. Remember that a high FSH level does not mean that you cannot get pregnant - it just means that your chances are dropping because your egg quality is impaired.
Some women find it difficult to understand why FSH levels are high in women with poor quality eggs. Intuitively, more is better, so higher levels should mean better eggs, shouldn't it ? As one patient asked me, " If FSH stands for Follicle Stimulating Hormone, and I have high levels of FSH, then doesn't that mean that I have the ability to stimulate lots of follicles ? A high FSH should mean that I should have lots of eggs ! "
I had to explain the basic biology to her. Normally , FSH is produced by the pituitary, and this is the hormone which is responsible for the growth of the egg from the ovary every month. In young women with lots of good quality eggs, low levels of FSH are enough to grow the eggs. However, as the woman grows older and egg quality and quantity decline, the pituitary needs to produce more and more FSH to stimulate egg growth, because the FSH has to work harder to stimulate egg growth.
Another test which has been recently developed is the measurement of the level of the hormone, inhibin B, in the blood. Low levels of inhibin B ( which are produced by " good " follicles) suggest a poor ovarian reserve. However, just because a test result is normal does not mean that the quality or number of the eggs produced will be good - the final proof of the pudding is always in the eating ! This is why one of the most useful ways of making a diagnosis of poor ovarian reserve is when the patient gives a history of responding poorly to medications used for superovulation in the past.
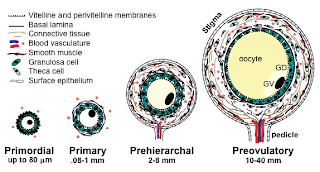
Along with using biochemical tests to assess ovarian function, we can use biophysical markers to test these too. These biophysical tests use ultrasound technology to image the ovaries and the follicles. The most useful test is called an antral follicle count ( AFC) , in which the doctor counts the number of antral follicles ( also referred to as resting follicles) present in the ovary on Day 3 using vaginal ultrasound scanning.
Antral follicles are small follicles , usually about 2-8 mm in diameter. The number of antral follicles correlates well with ovarian response. A normal total antral count is between 15 and 30. If the count is less than 6, the prognosis is poor. You can read more about the antral follicle count and see ultrasound images of these at the www.advancedfertility.com website . The volume of the ovaries also correlates with ovarian response. The volume of each ovary is calculated using the formula ( length × width × height × 0.5 ) and the normal ovarian volume of both ovaries combined is 10 ml. Women with small ovaries ( volume of less than 4 ml) have a poor ovarian response.
While an older woman often expects to have poor ovarian reserve, and is prepared for the fact that she may respond poorly to superovulation, when a young woman finds out she is a poor ovarian responder, this comes as a rude blow. Most young women expect that their eggs will be fine, because they are young and have regular cycles, but this is not always true. Regular periods simply means that the eggs are good enough to produce enough hormones to have normal menstrual cycles; however, this does not mean that the egg quality is good enough to make a baby !
Ovarian reserve is a biological variable, and egg quantity and quality in an individual woman can be average for her age, better than average, or worse than average. Women with poor egg quality are said to have poor ovarian reserve , poor ovarian function, or occult ovarian failure; and Dr Jansen has coined the term , oopause, to describe this condition.
This diagram by Dr Norbert Gleicher illustrates the concept of a "fertility curve," which suggests there may be a "norm" for ovarian aging, and hence cases in which ovarian aging can be termed "premature."
Many women cannot understand why they will have a problem producing lots of eggs when they have regular menstrual cycles. " If I produce an egg every month, then why can't I produce a lot of eggs when you give me a lot of HMG injections for superovulation ? " I explain that just because a car goes at 10 mph when you drive it in 1st gear, this does not mean you can predict what it's speed will be when you drive it in 4th gear !
Many treatment strategies have been developed in order to treat women with poor ovarian reserve. Because time is at a premium for these women, treatment needs to be aggressive, in order to help them conceive before their eggs run out completely. IVF is usually their best option, as it offers the highest success rates. Superovulating these women can be quite tricky, and this is where the experience and the expertise of the doctor makes a critical difference !
While it is true that a skilled doctor will be able to design an optimal superovulation for women with poor ovarian reserve, it is also true that the results are still likely to be poor. While Michael Schumacher will drive your car much better than you will ever be able to, if you give him a broken-down lemon to drive , even his skills are likely to let him down !
They usually need much higher doses of gonadotropin injections ( HMG) for superovulation. We have used upto 750 IU of HMG ( 10 amp of 75 IU) daily for difficult women, in order to stimulate them to grow eggs. Unfortunately, this is like scraping the bottom of the barrel, and the quantity and quality of their eggs often still remains poor.
Recently, we have had extremely good results with the Letrozole-Antagon protocol for poor ovarian responders.
In this protocol, downregulation is not needed, and we use your own gonadotropins to improve follicular recruitment in order to help you grow more eggs.
For patient who are poor ovarian responders, we transfer all the embryos we get, to maximise the chances of implantation. In this group, the risk of a multiple pregnancy is very low.
After the transfer, luteal phase support is provided with daily Progynova ( estradiol valerate2 mg, 3 tab daily; and Uterogestan ( 200 mg), 6 vaginal suppositories daily. You can travel back 3 days after the embryo transfer.
14 days after the transfer, you need to do a blood test for beta HCG to confirm a pregnancy.
Other clinics have tried using rec FSH ( recombinant gonadotropins) or GnRH antagonists, but neither of these help. In the past, doctors tried adding growth hormone injections ( because of the “growth factors” this contained) , but this was of no use. Interestingly, some doctors have gone back to using the natural cycle, or trying gentle stimulation with clomiphene for these women, since they don’t see any benefit in spending hundreds of dollars just to get 2-3 more eggs for IVF.
Interestingly, we see a lot of women who are iatrogenic poor ovarian responders – who have a poor ovarian response because they have been badly superovulated. These are typically women who have PCOD ( polycystic ovarian disease), who are undergoing IVF in clinics which don’t have much experience with treating such patients. Because their doctors are so scared of ovarian hyperstimulation syndrome ( OHSS), in their anxiety to prevent this complication, they often trigger off ovulation and egg retrieval too early.
As a result of this mis-timing of the HCG shot, most of the eggs retrieved are immature, and fail to fertilise. These woman are then labeled as being poor ovarian responders , when in reality it is their doctors who are poor ovarian stimulators ! If they are superovulated properly in a good IVF clinic, their pregnancy rates are excellent.
What happens if you are young and find that you have a poor ovarian response in the middle of your first IVF cycle ? This is a very difficult problem, because it was not anticipated, and you are not emotionally prepared to deal with it. Options include: continuing the cycle with an increased dose of injections; or canceling this cycle and starting a new cycle later with a higher dose of injections. However, the prognosis remains poor, and there is no certainty that you will grow more eggs with a higher dose the next time around.
The option which offers the highest pregnancy rate for women with a poor ovarian response is to use donor eggs. While this is medically straight forward, it can be very hard for a young woman with regular cycles to accept this option. Often, it’s worth doing one cycle with your own eggs even if the chances are poor, so that you have peace of mind that you did your best. This also may make it easier to explore the option of donor eggs for the future.
Source: https://www.drmalpani.com/articles/highfsh
TODAY'S BOOK SUGGESTION:
 It Starts with the Egg:
It Starts with the Egg:How the Science of Egg Quality Can Help You Get Pregnant Naturally, Prevent Miscarriage, and Improve Your Odds in IVF
by Rebecca Fett
-- Whether you are trying to conceive naturally or through IVF, the quality of your eggs will have a powerful impact on how long it takes you to get pregnant and whether you face an increased risk of miscarriage.
Poor egg quality is emerging as the single most important cause of age-related infertility, recurrent miscarriage, and failed IVF cycles. It is also a major contributor to infertility in PCOS.
Based on a comprehensive investigation of a vast array of scientific research, It Starts with the Egg reveals a groundbreaking new approach for improving egg quality and fertility.
With a concrete strategy including minimizing exposure to toxins such as BPA and phthalates, choosing the right vitamins and supplements to safeguard developing eggs, and harnessing nutritional advice shown to boost IVF success rates, this book offers practical solutions to will help you get pregnant faster and deliver a healthy baby.
Click to order/for more info: It Starts with the Egg
Don't have a Kindle? Get your Kindle here, or download a FREE Kindle Reading App.
 Abstract
AbstractBackground: Effects of androgens on follicle maturation have been controversial for some time.
Here, we review the potential of their applications in improving human ovulation induction, based on human and animal data, reported in the literature.
Discussion: Selective androgens appear capable of improving early stages of folliculogenesis.
They, therefore, may represent forerunners of a completely new class of ovulation-inducing medications, which, in contrast to gonadotropins, affect follicle maturation at much earlier stages.
Read rest of article
TODAY'S BOOK SUGGESTION
 Making Babies the Hard Way: Living with Infertility and Treatment
Making Babies the Hard Way: Living with Infertility and Treatmentby Caroline Gallup
-- A frank account of one couple's discovery that they cannot have children of their own, and their ensuing struggle through four years of fertility treatment.
One in six couples worldwide seek assistance to conceive and 80 per cent of couples undergoing fertility treatment are currently unsuccessful.
Writing with humour and honesty, Caroline Gallup describes the social, emotional, spiritual and physical impact of infertility on her and her husband, Bruce, including feelings of bereavement for the absent child, the unavoidable sense of inadequacy and the day-to-day difficulties of financial pressure.
As well as telling her own moving story, she also offers information and guidance for others who are infertile, or who are considering or undergoing treatment.
Click to order/for more info: Making Babies the Hard Way
Find it on Amazon: US | CDN | UK
Don't have a Kindle? Get your Kindle here, or download a FREE Kindle Reading App.
 Any woman who is struggling with fertility is likely to have dozens of numbers on her mind: bbt temps, sperm counts, follicle counts, uterine lining, estrogen, FSH, HCG, dosages of medications, age, numbers of cells, percents of morphology, success rates for clinics, success rates for procedures, days till ovulation, days till the next test... the list could go on and on.
Any woman who is struggling with fertility is likely to have dozens of numbers on her mind: bbt temps, sperm counts, follicle counts, uterine lining, estrogen, FSH, HCG, dosages of medications, age, numbers of cells, percents of morphology, success rates for clinics, success rates for procedures, days till ovulation, days till the next test... the list could go on and on.Unfortunately, all too often these numbers become an identity for what a woman believes is her future. I cannot say how often I’ve heard a woman summarize her situation by saying something like:
“Well, my FSH is 30.”
“But my luteal phase is only 8 days long.”
“The numbers aren’t doubling.”
“I’ve got a 10% chance with my own eggs.”
These statements seem like simple facts, but they often signify so much more: I’m afraid, I’m too old, I’ve waited too long, There’s something wrong with me, There’s nothing I can do, I’m waiting to have a miscarriage, My heart is breaking, I don’t think I’ll ever be a mother. Somehow, in some clinical way, the numbers make it easier to say.
Read more...
Life Begins... - Miscarriage stories of loss, hope & help
Pregnancy Stories by Age - Daily blog of hope & inspiration!
Stories of Pregnancy over 44 years old - sharing stories I find online, for inspiration!
Pregnancy Stories by Age - Daily blog of hope & inspiration!
Stories of Pregnancy over 44 years old - sharing stories I find online, for inspiration!
Recent Keyword Searches: menopause baby, what percentage of women age 34 can get pregnant?, keep your fertile in your 40s, how old can a woman be to get pregnant, change of life pregnancy
Photo by www.advancedfertility.com
 Posted by jbeskimo on the TTC in our Forties forum on FertilityFriend.com :
Posted by jbeskimo on the TTC in our Forties forum on FertilityFriend.com :
I don't know if this helps but I found this quote on an egg donor site:
Quote:
For example, we see some women in their early 20's that want to be egg donors at our center that when we do ultrasound on them we see only 3 antral follicles per ovary - and the volume of the ovary is low. Although we no longer accept these women into our egg donor program, I am not convinced that they have an egg quality or quantity problem (my best guess is that they have reduced quantity, but quality is probably fine). Often the young woman is concerned about her future fertility when we tell them that their ovaries are small and do not look like they will make sufficient eggs to be a good donor candidate.
However, I am not aware of any data (medical research, etc.) that shows that women in that particular scenario are going to have a fertility problem. They might - but we just don't know. Some of those women already have 3 kids and are more fertile than they would prefer - so we really can't say that all women with low antral follicle counts have a fertility problem (reduced egg quality or quantity).
Source: http://www.advancedfertility.com/antralfollicles.htm
Just remember that the field of fertility is still very nebulous.
Source: http://www.fertilityfriend.com/Circles/viewtopic.php?p=19403620#19403620
Life Begins...
Miscarriage stories of loss, hope & help
http://born2luv.blogspot.com/
Stories of Pregnancy & Birth over 44y
- Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
4,650 Stories of Pregnancy & Birth over 44y
Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
Recent Keyword Searches: wanting to get pregnant at 40, omega 3 be taken to get pregnant, do you need estrogen to get pregnant, how to get healthy to get pregnant at 40yrs, can repeated orgasms help you get pregnant during ovulation
 Posted by jbeskimo on the TTC in our Forties forum on FertilityFriend.com :
Posted by jbeskimo on the TTC in our Forties forum on FertilityFriend.com :I don't know if this helps but I found this quote on an egg donor site:
Quote:
For example, we see some women in their early 20's that want to be egg donors at our center that when we do ultrasound on them we see only 3 antral follicles per ovary - and the volume of the ovary is low. Although we no longer accept these women into our egg donor program, I am not convinced that they have an egg quality or quantity problem (my best guess is that they have reduced quantity, but quality is probably fine). Often the young woman is concerned about her future fertility when we tell them that their ovaries are small and do not look like they will make sufficient eggs to be a good donor candidate.
However, I am not aware of any data (medical research, etc.) that shows that women in that particular scenario are going to have a fertility problem. They might - but we just don't know. Some of those women already have 3 kids and are more fertile than they would prefer - so we really can't say that all women with low antral follicle counts have a fertility problem (reduced egg quality or quantity).
Source: http://www.advancedfertility.com/antralfollicles.htm
Just remember that the field of fertility is still very nebulous.
Source: http://www.fertilityfriend.com/Circles/viewtopic.php?p=19403620#19403620
Life Begins...
Miscarriage stories of loss, hope & help
http://born2luv.blogspot.com/
Stories of Pregnancy & Birth over 44y
- Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
4,650 Stories of Pregnancy & Birth over 44y
Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
Recent Keyword Searches: wanting to get pregnant at 40, omega 3 be taken to get pregnant, do you need estrogen to get pregnant, how to get healthy to get pregnant at 40yrs, can repeated orgasms help you get pregnant during ovulation
 Posted on FertilityFriend.com by Babycakes2007:
Posted on FertilityFriend.com by Babycakes2007:Hi, Ladies! I'm visiting from the IVF board but wanted to post this here at well. This study is open to women ages 18-45 so I wanted to make sure as many people know about it as possible. These IVF studies fill up quickly!!
I was cruising the clinicaltrails.gov website and found an open IVF study at the University of California in San Francisco. I thought I'd pass it along to anyone in the Bay area who was interested. I'm in a research study in Florida and it's the best thing that happened to us. It's saving us $11,000!! I believe in "paying it forward" so here you go! Good luck!!
Here is the Inclusion Criteria for the study:
Ages Eligible for Study: 18 Years to 45 Years
Infertile patients
Undergoing IVF
Antral Follicle Count Less than 10
Prior history of less than 5 oocytes generated during an IVF cycle
Here is the link-
http://clinicaltrials.gov/ct2/show/NCT00334243
Search for more studies:
Search for Clinical Trials
Find trials for a specific medical condition or other criteria in the ClinicalTrials.gov registry. ClinicalTrials.gov currently has 56,865 trials with locations in 156 countries.
Find trials for a specific medical condition or other criteria in the ClinicalTrials.gov registry. ClinicalTrials.gov currently has 56,865 trials with locations in 156 countries.
Life Begins...
Miscarriage stories of loss, hope & help
http://born2luv.blogspot.com/
Stories of Pregnancy & Birth over 44y
- Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
4,600 Stories of Pregnancy & Birth over 44y
Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
Photo by www.dailymail.co.uk
 A major breakthrough by British scientists could bring new hope for women facing the heartbreak of infertility.
A major breakthrough by British scientists could bring new hope for women facing the heartbreak of infertility.
For the first time a team has managed to grow hundreds of eggs in the laboratory using a new technique which could help cancer patients whose treatment can leave them infertile.
It would also allow thousands more women to wait until middle age to have children.
The scientists from Edinburgh University have shown that immature eggs can be frozen, grown and matured in the lab.
The process could lead to women having pieces of ovary containing the immature eggs removed and stored. Much later on, they could be thawed, fertilised and finally implanted into the womb.
The breakthrough, which comes as MPs prepare to debate the controversial Human Fertilisation and Embryology Bill, raises again the ethical debate surrounding the freezing of eggs.
Some say it is morally wrong for a woman to do so and have them fertilised years later in order to delay having children while she pursues a career.
However, scientists hope the new process will revolutionise fertility preservation for women because it will allow them to store many more eggs than they can under traditional IVF techniques.
And, because immature eggs survive the freezing process much better than the mature ones used in IVF, it is much more likely that older women will be able to conceive using them.
It brings forward the prospect that thousands of women will use the technique to side-step the menopause, delaying motherhood for the sake of their careers.
The process, which could be available in five years, also provides hope for cancer sufferers who at the moment are often left infertile following chemotherapy and radiotherapy.
Powerful anti-cancer drugs can destroy follicles in the ovaries, wiping out any possibility of women having children.
At the moment, these women have a piece of ovary removed, frozen and then re-transplanted after their cancer treatment. But there is always the danger that the cancer could be reintroduced by the implant.
The new technique means women's immature eggs contained in the patch of ovary could be grown in the lab and then screened for cancer before being used in IVF treatment.
There are also hopes it will provide a rich source of eggs for scientists to study for clues on ways to treat a range of diseases.
There is a shortage of human eggs for medical research, and if the technique works it would bypass the controversy over the use of animal-human 'hybrid' embryos.
The Human Fertilisation and Embryology Bill, which MPs are due to debate next month, would allow scientists to use these embryos in medical research, but many MPs plan to vote against.
The research, carried out by an Edinburgh University team led by Dr Evelyn Telfer, is published today in the journal Human Reproduction.
"This is a significant step in developing immature eggs to maturity outside the body," said Dr Telfer.
"Women who face infertility as a result of chemotherapy, or who want to put their biological clock on hold, could benefit from this system.
"However, there is a lot more research to be carried out before this technique could be safely applied within a clinical setting."
Last year, Canadian scientists announced the first birth of a child created from a human egg matured in the laboratory.
However, they did not use the same "primordial follicles" studied by the researchers from Edinburgh.
These are the tiny egg-bearing pockets within the ovaries that are present in their millions at birth, but gradually die off over the course of a woman's life.
They represent a woman's fertility "battery" which once depleted cannot be recharged.
Many remain dormant, but some go on to mature and eventually release their eggs in preparation for fertilisation.
For the first time, the team led by Dr Telfer has succeeded in growing primordial follicles to a late stage of maturation in the lab.
They took pieces of ovary containing the follicles from six volunteer women who were giving birth by caesarean section.
These were then exposed to a chemical that promotes growth, similar to the one that functions in the ovaries.
Around a third of them survived and went on to reach the advanced 'antral' stage of development.
At this stage, the follicles are filled with fluid and contain eggs almost ready to be fertilised.
It means that it could soon be possible to grow hundreds of eggs in the laboratory.
The Canadian team which managed to create a child from immature eggs was working with only around a dozen.
Another advantage is that the follicles mature much more quickly in the lab than they do in the ovary.
The scientists do not yet know whether eggs - or oocytes, to give them their technical name - matured in this way are completely normal and suitable for in-vitro fertilisation. But animal studies suggest they are.
The next step is to use hormones and other substances to try to nudge the 'antral' eggs on to the next stage of maturation, and then test the technique on humans.
"We believe there's good evidence that we can get normal oocytes, but of course you would never apply this technique clinically until you are sure," said Dr Telfer.
"We're seeking funding for further research to bridge that gap. It might take five to ten years from now before we get to the stage of a clinical trial."
Dr Jane Stewart, consultant in reproductive medicine at the Newcastle Fertility Centre, said:
"This work increases our understanding of the maturation of human eggs in the lab and takes us a step nearer the goal of strong immature eggs for fertility preservation for women."
Source: http://www.dailymail.co.uk/pages/live/articles/health/healthmain.html?in_article_id=560951&in_page_id=1774
Life Begins...
Miscarriage stories of loss, hope & help
http://born2luv.blogspot.com/
Stories of Pregnancy & Birth over 44y
- Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
4,600 Stories of Pregnancy & Birth over 44y
Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
 A major breakthrough by British scientists could bring new hope for women facing the heartbreak of infertility.
A major breakthrough by British scientists could bring new hope for women facing the heartbreak of infertility.For the first time a team has managed to grow hundreds of eggs in the laboratory using a new technique which could help cancer patients whose treatment can leave them infertile.
It would also allow thousands more women to wait until middle age to have children.
The scientists from Edinburgh University have shown that immature eggs can be frozen, grown and matured in the lab.
The process could lead to women having pieces of ovary containing the immature eggs removed and stored. Much later on, they could be thawed, fertilised and finally implanted into the womb.
The breakthrough, which comes as MPs prepare to debate the controversial Human Fertilisation and Embryology Bill, raises again the ethical debate surrounding the freezing of eggs.
Some say it is morally wrong for a woman to do so and have them fertilised years later in order to delay having children while she pursues a career.
However, scientists hope the new process will revolutionise fertility preservation for women because it will allow them to store many more eggs than they can under traditional IVF techniques.
And, because immature eggs survive the freezing process much better than the mature ones used in IVF, it is much more likely that older women will be able to conceive using them.
It brings forward the prospect that thousands of women will use the technique to side-step the menopause, delaying motherhood for the sake of their careers.
The process, which could be available in five years, also provides hope for cancer sufferers who at the moment are often left infertile following chemotherapy and radiotherapy.
Powerful anti-cancer drugs can destroy follicles in the ovaries, wiping out any possibility of women having children.
At the moment, these women have a piece of ovary removed, frozen and then re-transplanted after their cancer treatment. But there is always the danger that the cancer could be reintroduced by the implant.
The new technique means women's immature eggs contained in the patch of ovary could be grown in the lab and then screened for cancer before being used in IVF treatment.
There are also hopes it will provide a rich source of eggs for scientists to study for clues on ways to treat a range of diseases.
There is a shortage of human eggs for medical research, and if the technique works it would bypass the controversy over the use of animal-human 'hybrid' embryos.
The Human Fertilisation and Embryology Bill, which MPs are due to debate next month, would allow scientists to use these embryos in medical research, but many MPs plan to vote against.
The research, carried out by an Edinburgh University team led by Dr Evelyn Telfer, is published today in the journal Human Reproduction.
"This is a significant step in developing immature eggs to maturity outside the body," said Dr Telfer.
"Women who face infertility as a result of chemotherapy, or who want to put their biological clock on hold, could benefit from this system.
"However, there is a lot more research to be carried out before this technique could be safely applied within a clinical setting."
Last year, Canadian scientists announced the first birth of a child created from a human egg matured in the laboratory.
However, they did not use the same "primordial follicles" studied by the researchers from Edinburgh.
These are the tiny egg-bearing pockets within the ovaries that are present in their millions at birth, but gradually die off over the course of a woman's life.
They represent a woman's fertility "battery" which once depleted cannot be recharged.
Many remain dormant, but some go on to mature and eventually release their eggs in preparation for fertilisation.
For the first time, the team led by Dr Telfer has succeeded in growing primordial follicles to a late stage of maturation in the lab.
They took pieces of ovary containing the follicles from six volunteer women who were giving birth by caesarean section.
These were then exposed to a chemical that promotes growth, similar to the one that functions in the ovaries.
Around a third of them survived and went on to reach the advanced 'antral' stage of development.
At this stage, the follicles are filled with fluid and contain eggs almost ready to be fertilised.
It means that it could soon be possible to grow hundreds of eggs in the laboratory.
The Canadian team which managed to create a child from immature eggs was working with only around a dozen.
Another advantage is that the follicles mature much more quickly in the lab than they do in the ovary.
The scientists do not yet know whether eggs - or oocytes, to give them their technical name - matured in this way are completely normal and suitable for in-vitro fertilisation. But animal studies suggest they are.
The next step is to use hormones and other substances to try to nudge the 'antral' eggs on to the next stage of maturation, and then test the technique on humans.
"We believe there's good evidence that we can get normal oocytes, but of course you would never apply this technique clinically until you are sure," said Dr Telfer.
"We're seeking funding for further research to bridge that gap. It might take five to ten years from now before we get to the stage of a clinical trial."
Dr Jane Stewart, consultant in reproductive medicine at the Newcastle Fertility Centre, said:
"This work increases our understanding of the maturation of human eggs in the lab and takes us a step nearer the goal of strong immature eggs for fertility preservation for women."
Source: http://www.dailymail.co.uk/pages/live/articles/health/healthmain.html?in_article_id=560951&in_page_id=1774
Life Begins...
Miscarriage stories of loss, hope & help
http://born2luv.blogspot.com/
Stories of Pregnancy & Birth over 44y
- Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
4,600 Stories of Pregnancy & Birth over 44y
Daily blog of hope & inspiration!
http://pregnancyover44y.blogspot.com/
Purpose
Our long term goal is to elucidate the role of DHEA on follicular dynamics in the human ovary and to better understand the interaction of DHEA supplementation with other treatments for ovulation induction, especially among older reproductive age women.
The specific hypothesis behind the proposed research is that DHEA is a regulator of follicular dynamics acting in the early pre-gonadotropin dependent stage of initial primordial follicle recruitment and growth.
* Clinical Pregnancy per cycle of treatment
Secondary Outcome Measures:
* Embryos per cycle of treatment
* Oocytes per cycle of treatment
Total Enrollment: 200
Study start: January 2007
Specific Aims:
Our long term goal is to elucidate the role of DHEA on follicular dynamics in the human ovary and to better understand the interaction of DHEA supplementation with other treatments for ovulation induction, especially among older reproductive age women.
Based on these observations the experimental focus of this project is on the interaction of DHEA adjuvant treatment with gonadotropin treatment during ovulation induction. The specific aims are designed to provide a comprehensive assessment of this effect.
1. DHEA treatment will increase pregnancy rates among women 40 to 45 years old.
2. DHEA treatment will increase antral follicle counts.
3. DHEA will lead to increased anti-mullerian (AMH) hormone levels.
4. DHEA treatment will increase mean and peak follicular phase estradiol.
5. DHEA treatment will increase the number of oocytes retrieved in IVF cycles compared to placebo.
Research Plan:
Placebo controlled randomized trial of 2 months of DHEA pretreatment prior to ovulation induction for IVF-ET.
Hypothesis 1:
Two months of DHEA pretreatment will improve the chance of pregnancy and lead to greater oocyte and embryo yields relative to placebo treated control patients.
Hypothesis 2:
Embryos produced following 2 months of pretreatment with either DHEA alone OR with DHEA plus supplemental FSH 150 units per day will result in embryos with better morphology
Hypothesis 3a-c:
Two months of DHEA pretreatment will result in:
a) increased antral follicle counts, increased AMH,
b) increased mean and peak estradiol and
c) increased oocyte production
More Info: http://clinicaltrials.gov/ct/show/NCT00419913?order=4
Our long term goal is to elucidate the role of DHEA on follicular dynamics in the human ovary and to better understand the interaction of DHEA supplementation with other treatments for ovulation induction, especially among older reproductive age women.
The specific hypothesis behind the proposed research is that DHEA is a regulator of follicular dynamics acting in the early pre-gonadotropin dependent stage of initial primordial follicle recruitment and growth.
* Clinical Pregnancy per cycle of treatment
Secondary Outcome Measures:
* Embryos per cycle of treatment
* Oocytes per cycle of treatment
Total Enrollment: 200
Study start: January 2007
Specific Aims:
Our long term goal is to elucidate the role of DHEA on follicular dynamics in the human ovary and to better understand the interaction of DHEA supplementation with other treatments for ovulation induction, especially among older reproductive age women.
Based on these observations the experimental focus of this project is on the interaction of DHEA adjuvant treatment with gonadotropin treatment during ovulation induction. The specific aims are designed to provide a comprehensive assessment of this effect.
1. DHEA treatment will increase pregnancy rates among women 40 to 45 years old.
2. DHEA treatment will increase antral follicle counts.
3. DHEA will lead to increased anti-mullerian (AMH) hormone levels.
4. DHEA treatment will increase mean and peak follicular phase estradiol.
5. DHEA treatment will increase the number of oocytes retrieved in IVF cycles compared to placebo.
Research Plan:
Placebo controlled randomized trial of 2 months of DHEA pretreatment prior to ovulation induction for IVF-ET.
Hypothesis 1:
Two months of DHEA pretreatment will improve the chance of pregnancy and lead to greater oocyte and embryo yields relative to placebo treated control patients.
Hypothesis 2:
Embryos produced following 2 months of pretreatment with either DHEA alone OR with DHEA plus supplemental FSH 150 units per day will result in embryos with better morphology
Hypothesis 3a-c:
Two months of DHEA pretreatment will result in:
a) increased antral follicle counts, increased AMH,
b) increased mean and peak estradiol and
c) increased oocyte production
More Info: http://clinicaltrials.gov/ct/show/NCT00419913?order=4
OBJECTIVE: The objective of this study was 2-fold. The first was to estimate side-to-side variation in antral follicle counts. The second was to determine whether basal follicle-stimulating hormone levels on days 2, 3, and 4 of the same menstrual cycle are significantly different.
METHODS: Forty-one patients between the ages of 20 and 42 years undergoing monitoring for in vitro fertilization-embryo transfer were evaluated ultrasonographically for antral follicle number. The antral follicle counts were determined for each ovary by experienced ultrasonographers at the time of suppression check ultrasonography. In a separate study, 62 normal subjects (ages 20–25 and 40–45 years) underwent serial sequential serum follicle-stimulating hormone determinations on days 2–4 of the menstrual cycle, and these levels were compared.
RESULTS: There was no significant difference between right and left antral follicle counts (P = .30). Serial follicle-stimulating hormone values were not significantly different on days 2, 3, or 4 of the menstrual cycle (P = .22).
CONCLUSION: There is no significant difference between right-sided and left-sided antral follicle counts within the same individual. In turn, there is no significant difference in serial follicle-stimulating hormone levels on days 2, 3, or 4 of the menstrual cycle.
Full article: http://www.greenjournal.org/cgi/content/full/104/4/801
METHODS: Forty-one patients between the ages of 20 and 42 years undergoing monitoring for in vitro fertilization-embryo transfer were evaluated ultrasonographically for antral follicle number. The antral follicle counts were determined for each ovary by experienced ultrasonographers at the time of suppression check ultrasonography. In a separate study, 62 normal subjects (ages 20–25 and 40–45 years) underwent serial sequential serum follicle-stimulating hormone determinations on days 2–4 of the menstrual cycle, and these levels were compared.
RESULTS: There was no significant difference between right and left antral follicle counts (P = .30). Serial follicle-stimulating hormone values were not significantly different on days 2, 3, or 4 of the menstrual cycle (P = .22).
CONCLUSION: There is no significant difference between right-sided and left-sided antral follicle counts within the same individual. In turn, there is no significant difference in serial follicle-stimulating hormone levels on days 2, 3, or 4 of the menstrual cycle.
Full article: http://www.greenjournal.org/cgi/content/full/104/4/801
Popular Posts
-
Found this on Women Over 40 With High FSH : TheFertilitycure.com -- I am sure many people here are very familiar with Randine Lewis...
-
Newborn baby , by Joelle Inge-Messerschmidt/Photographybyjoelle.com In the Infertility Support forum on TCOYF's [Taking Care of You...
-
Photo credit: Tea Cup , by Saxon ABSTRACT OBJECTIVES: The effect of caffeine consumption on fertility was examined prospectively in 210...
-
Photo credit: Yvonne , by Duane J A woman who is over age 43 or 44 years old, will often be turned away from any chance at assisted repr...
-
If getting pregnant is high on your list of resolutions for 2010, be sure to read this quick guide for conceiving and having a healthy baby ...
-
Menopause is the time in a woman's life when her reproductive system shuts down and her reproducing days are over... or are they? For s...










Recent Comments